Global API supplier and CDMO partner
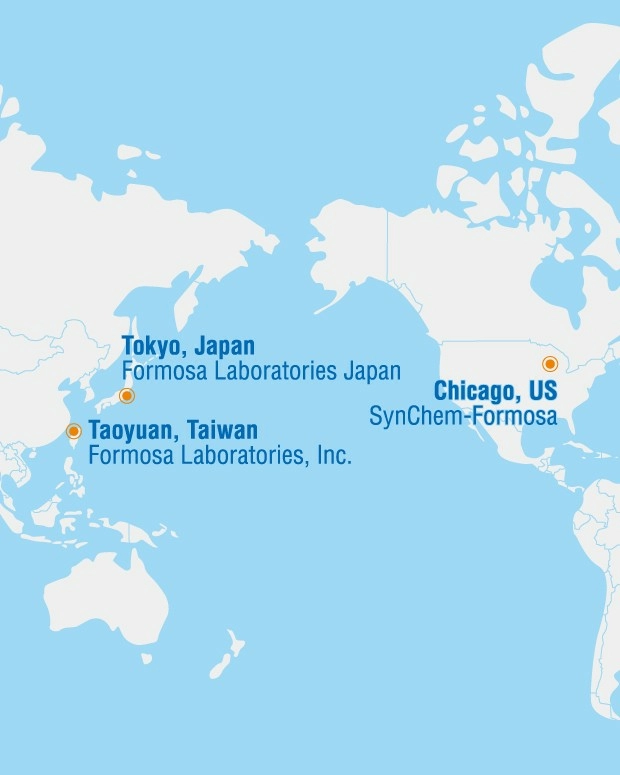
Formosa Laboratories was established as a CRO in 1995 to provide API process development, impurity identification and synthesis services, and entered GMP API manufacturing in 2000. Formosa Laboratories is located in Luzhu, Taoyuan, Taiwan, employs more than 900 people and occupies an area of 69,036 square meters. Formosa Laboratories has a wide range of manufacturing facilities, including API, High Potency API (HPAPI), Antibody-Drug conjugates (ADCs), and injectable. From the pilot plant to kilogram and tons scale manufacturing, Formosa Laboratories is well equipped with a wide range of equipment to meet the needs of different manufacturing processes and capacity to provide high quality API and Injectable.
Corporate Group
Company Video

FORMOSA
FORMOSA
Strategic CDMO partner for your success
Formosa Laboratories owns dozens of API products that are marketed globally, including vitamin D derivatives and cholesterol phosphate conjugate series that are world-leading in terms of quality and sales results; Formosa Laboratories also provides CDMO services including development and manufacturing services for, API, peptide, injectables and Antibody-Drug conjugates (ADC). The vertical integrated one-stop shop CDMO services allow us to meet clients' demand on time with outstanding quality.
Quality
Formosa Laboratories is committed to developing and manufacturing high quality products. We strive to produce the highest quality APIs and injectables in compliance with regulatory need. We owned ISO certification and passed GMP inspections by the Food and Drug Administration (FDA) of Taiwan's Ministry of Health and Welfare (MOHW), the U.S. FDA, Germany's BGV, the European Union's EDQM, and Japan's PMDA. Under the requirements of cGMP, Formosa Laboratories has been continuously updating and implementing its products to meet the current standards of Good Manufacturing Practice (GMP), and delivers its API products, injectables, and expands CDMO business to Europe, the United States, Japan, and the rest of the world.

Mission Statement
Hope, Passion, Innovation, Sustainability
- We develop and manufacture APIs, ADCs, and injectables with state-of-the-art technology and in compliance with the cGMP regulations.
- We provide comprehensive custom synthesis services efficiency and confidentiality.
- We build mutually beneficial relationship with our business partners
- We emphasize employee training to develop a highly motivated workforce.
- We ensure the safety in our workplace and minimize our negative impact to the environment.


